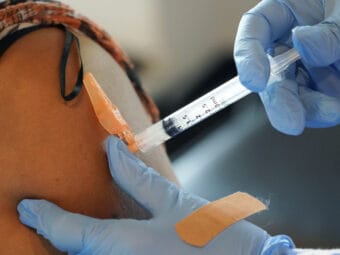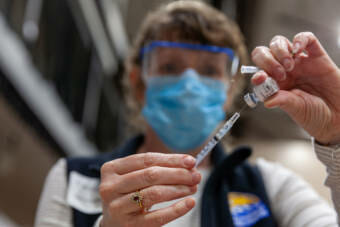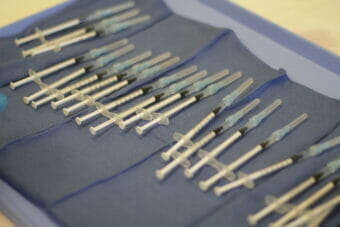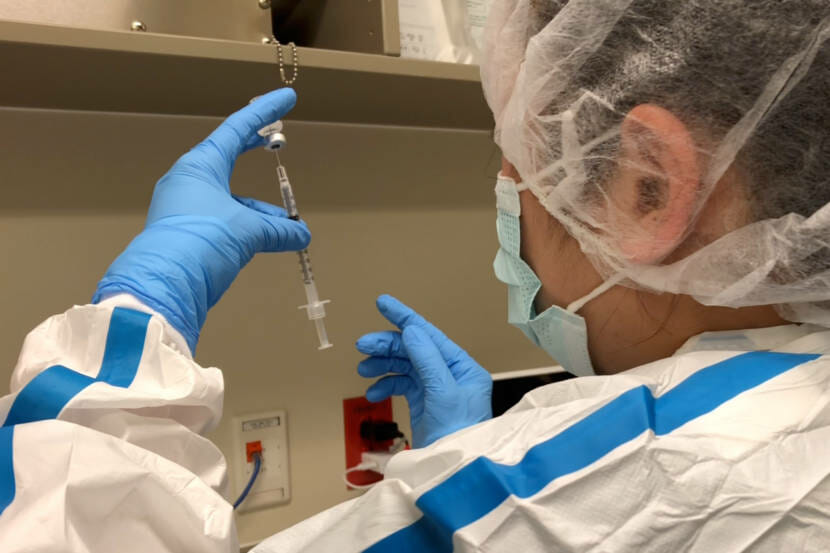
As providers at the Alaska Native Medical Center began injecting workers with the new COVID-19 vaccine from Pfizer and BioNTech this week, they started noticing something surprising: leftover liquid in each five-dose vial, sometimes enough for a full extra dose or even two.
“It was one of those hot topics,” said Kara King, the Anchorage hospital’s director of pharmacy. “Word was starting to spread that there were extra doses.”
The discovery was simultaneously thrilling and confounding, presenting providers with an array of questions: Were they doing the injections correctly? Could they use the extra vaccine? If they did, would there be enough in their next shipment for each recipient to get their required second injection?
Pharmacists around the country were making similar observations. And on Wednesday, the Food and Drug Administration issued a statement saying that “given the public health emergency,” it’s acceptable for providers to use “every dose obtainable” — though the agency cautioned against pooling leftover vaccine from multiple vials.
It’s not yet clear how many extra vaccines can now be administered, collectively, from ANMC’s total allotment, King said.
The bonus doses are also likely to present logistical headaches for the officials charged with distributing and tracking the scarce vaccine. But it’s a great problem to have, King added.
Alaska’s initial shipment of the Pfizer vaccine was expected to be enough for just 35,000 doses — only about 5% of the state’s population. Front-line health care workers, first responders providing medical care and residents and staff at nursing homes are among those prioritized in the first round of vaccinations.
“This is a super-scarce resource right now,” King said. “So, it is just really fantastic that we could potentially get more doses out of the vial and get more people vaccinated.”
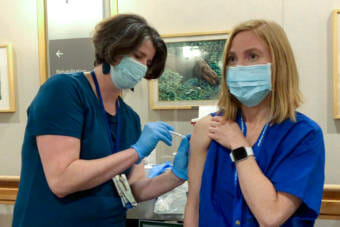
At least one other Anchorage hospital, Providence Alaska Medical Center, said it had also discovered extra doses in its Pfizer vials.
Pfizer told the New York Times that there’s the same amount of vaccine in each vial. But the amount left over after each dose could be different depending on the type of needles, syringes and diluting solutions used by providers, it said.
It told the newspaper that it was studying the issue with the FDA and couldn’t make its own recommendations about what to do with the extra vaccine.
Officials at the Alaska Department of Health and Social Services, which is leading the state’s efforts to distribute the vaccine, did not respond to a request for information about the extra doses.
But King said she’d reviewed a draft statement from the state that sounded similar to Pfizer’s guidance. The draft statement describes how the amount of vaccine drawn from each vial, for each dose, depends on the size of the needle being used, King said.
“And so if you can guarantee that the needle size you use for the first dose is the same needle size for the second dose, then you should be able to pull the same amount of vaccine out of every vial,” King said.
This is a breaking news story and will be updated.